Surgical Instruments For Arthroplasty
The demand for dependable surgical instruments for arthroplasty is rising across Southern Africa as hospitals standardize joint-reconstruction pathways and administrators push for predictable outcomes. That mix of modernization and tradition creates an opportunity for partners who can pair solid implants with instruments that are intuitive, durable, and easy to teach. Ortonom Medical keeps a narrow clinical focus so your hospital conversations stay practical: one coherent system, clear technique, and steady after-sales. When your local access meets our manufacturing discipline, trust builds quickly.
Clinical teams need more than a catalog; they need a pathway. With rotating staff, visiting surgeons, and busy theaters, the right surgical instruments for arthroplasty reduce anxiety because they make the sequence obvious and the checks repeatable. Color-coded blocks, labeled handles, and logical trays let new nurses orient fast and let seasoned scrub techs run the room without drama. That quiet reliability is what committees remember long after a tender is closed.
Procurement leaders care deeply about documentation. Instruments that arrive with clean IFUs, tray checklists, and sterilization instructions shorten approval time and help avoid costly delays. Ortonom Medical treats paperwork as part of the product, not an afterthought, so your team can submit once and move forward. In markets where face-to-face relationships remain decisive, the ability to back a promise with evidence and process separates a vendor from a true partner.
Finally, the traditional ethic—do what you said you would do—still wins. Honest lead times, conservative claims, and fast follow-through convert first cases into long-term relationships. The platform may be modern, but the craft is timeless: get the basics right, keep the trays ready, and stay present in the OR. That is how surgical instruments for arthroplasty become a franchise, not a one-off sale.
What Ortonom Medical Brings to Southern African Hospitals
Ortonom Medical builds implant families and instrument sets as a single ecosystem. For knee arthroplasty, the femoral component, tibial baseplate, polyethylene insert, and patella are validated to work together, and the instruments are sequenced to make each resection and trial step unambiguous. This coherence makes surgical instruments for arthroplasty teachable: if you learn one case, you can repeat the flow on the next list without surprises.
Documentation is purpose-built. Technical files, labels, and IFUs are formatted for hospital committees, and tray checklists map to sterilization pathways. That means your regulatory manager spends less time reformatting and more time advancing approvals. With fewer revisions and faster sign-offs, your first cases start earlier and your reputation grows faster.
Enablement is continuous. We provide bilingual (EN/FR) slides, short technique videos, and pocket checklists that scrub teams can use between cases. Launch workshops are practical and conservative: verification steps, common pitfalls, and how to recover calmly when something does not line up. After the first cases, we capture insert-thickness preferences, patella strategy, and instrument wear patterns and translate those signals into a smarter stock matrix.
Service is where promises become proof. Instrument turnaround times, spare availability, and rapid troubleshooting determine whether a strong first week becomes a stable first year. We design surgical instruments for arthroplasty programs around reliability: planned maintenance with CSSD, consignment for fast movers, and simple dashboards that show where every tray is in the cycle.
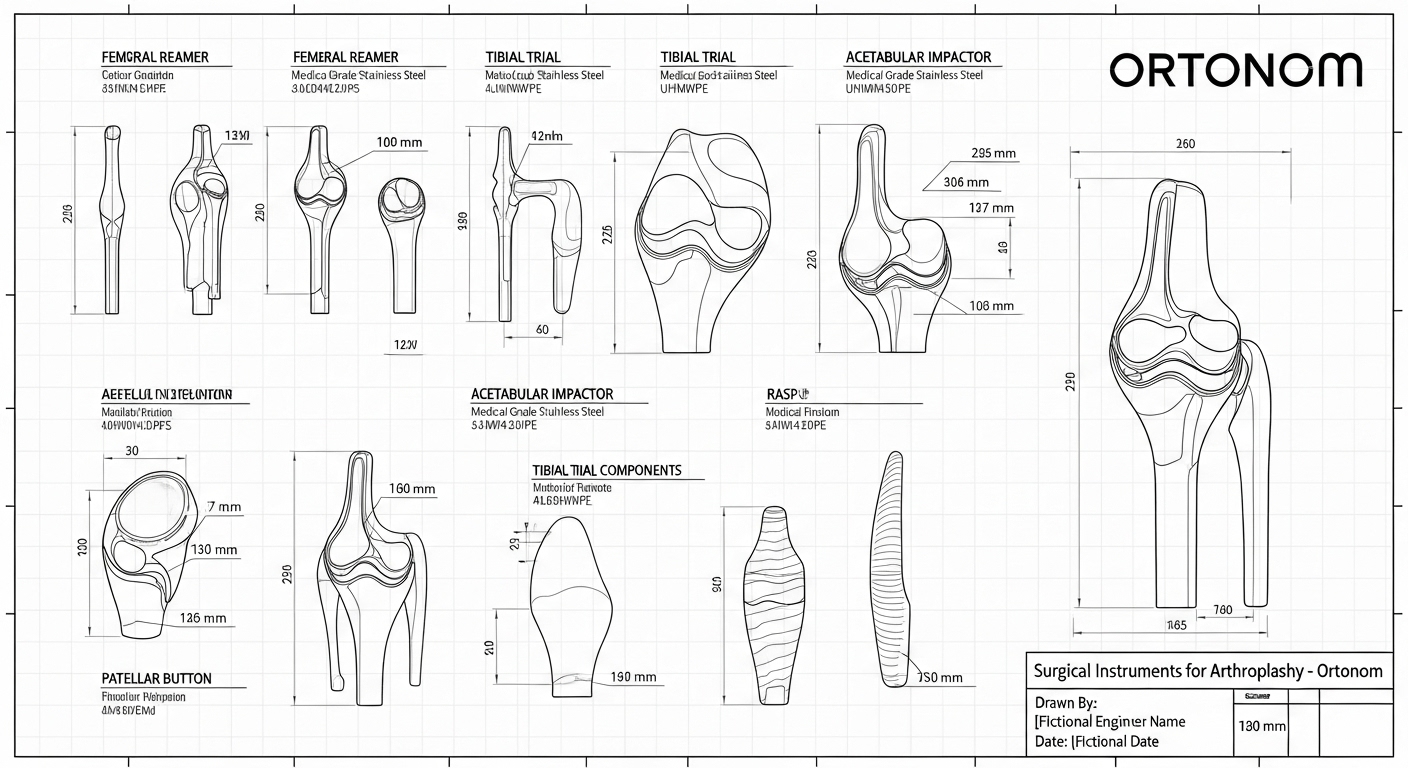
Inside the Kits: Architecture of Surgical Instruments for Arthroplasty
A great implant cannot shine without the right tools. Our surgical instruments for arthroplasty sets emphasize alignment accuracy, repeatable bone cuts, and intuitive trialing. Handles, guides, and blocks are labeled to match the operative sequence so teams can move without hesitation, and trays are laid out to minimize hand-offs and lost time.
Femoral preparation tools focus on consistent distal and posterior resections. Clear posterior-condyle references and trochlear alignment cues help surgeons preserve what is healthy while standardizing what varies. Tibial preparation tools support accurate slope and rotation, with anatomic footprints that reduce overhang risk—one of the common causes of soft-tissue irritation.
Trial components make gap assessment visible. Insert trials step in predictable thickness increments so balancing feels like a decision, not a guess. The locking interface is robust yet serviceable, allowing quick exchanges without stressing the scrub flow. Patellar instruments support both dome and anatomical strategies, with depth stops that protect bone and tracking checks that reduce rework.
Sterilization and durability matter as much as ergonomics. Surfaces resist scratching during trial exchanges, and trays are designed to dry efficiently, reducing turnaround times. When hospitals see that your surgical instruments for arthroplasty keep their accuracy after many cycles, they associate your brand with calm, orderly theaters—a classic reputation that opens doors.
Winning the OR: Technique, Training, and Teachability
Hospitals buy confidence as much as components. That is why we center workshops on the moments that decide outcomes: sequencing of cuts, verification points, and gap logic. Short video modules reinforce the same steps so rotating staff can refresh in minutes. When instruction and instruments tell the same story, surgical instruments for arthroplasty become easy to adopt at scale.
We encourage “pause points” that respect surgical tradition: confirm distal femoral resection before moving on; re-check tibial slope and rotation after pinning; verify trial tracking with patella before final implant. These pauses feel old-school, but they prevent downstream corrections and instrument wear. Over time, they shorten lists because they reduce surprises.
Post-launch, we support supervised first cases and structured debriefs. If a surgeon prefers a certain insert thickness range or a different patella resurfacing plan, we capture it and adjust the starter matrix. That makes your stock honest—aligned to real cases, not optimistic forecasts. It also improves morale: theaters run smoothly when trays match habits.
Teachability protects margins. Stable operative times reduce overtime, fewer emergency shipments protect cash, and consistent instrument condition lowers refurbishment costs. In tender rooms, this adds up to a simple message: our surgical instruments for arthroplasty make life easier for surgeons, nurses, sterilization teams, and finance.
Country Opportunity Snapshot – Southern Africa Focus

Use the table below to plan sequencing, tailor training calendars, and shape stock matrices for Southern Africa. Keep the core story consistent, then localize language, tender norms, and logistics corridors.
| Country | Business Languages | First Target Cities | Dominant Channels | Distributor Profile Fit |
|---|---|---|---|---|
| South Africa | English Afrikaans | Johannesburg Cape Town Durban | Private + public tenders | Clinical education strength strong CSSD ties |
| Namibia | English | Windhoek | Private hospitals | Inventory discipline service SLAs |
| Botswana | English | Gaborone Francistown | Private + NGO | Agile import dependable after-sales |
| Zimbabwe | English | Harare Bulawayo | Private hospitals | Training focus OR presence |
| Zambia | English | Lusaka Ndola | Public + private | Tender literacy hospital relationships |
| Mozambique | Portuguese | Maputo Beira | Public | Documentation rigor bilingual assets |
| Angola | Portuguese | Luanda | Private + public | Capital equipment experience logistics depth |
| Malawi | English | Lilongwe Blantyre | NGO + private | Education capability fast field support |
| Lesotho | English | Maseru | Public | Dossier management committee access |
| Eswatini | English | Mbabane Manzini | Private | Reliable service quick troubleshooting |
Start with two lighthouse sites—one higher-volume metro (e.g., Johannesburg or Cape Town) and one neighboring market with fast decision cycles (e.g., Windhoek or Gaborone). Ten to twelve clean cases per site, with outcomes summarized in bilingual briefs, establish credibility. Then expand to adjacent countries that share language or logistics lanes so you can reuse training and documents with minimal friction.
Stakeholder maps are essential. List the chief surgeon, nursing lead, procurement officer, CSSD manager, and finance contact for each hospital. Record their constraints and what “good” looks like to them. Those notes become local copies of IFUs, checklists, and brochures. In committee rooms, this preparation reads as respect—and it moves decisions faster.
Bilingual assets win time. EN materials cover most of Southern Africa, while Portuguese assets are decisive in Angola and Mozambique. Pair slide decks with concise one-page “spec snapshots” that show tray contents, sizes, and maintenance cadence. When people see process, they trust the promise.
Compliance Logistics and After-Sales SLAs
Procurement leaders want two outcomes above all: files that pass on the first try and trays that arrive when promised. The table clarifies responsibilities so public claims match day-to-day reality.
| Item | Ortonom Medical Provides | Partner Commits | Typical Timeline |
|---|---|---|---|
| Regulatory dossier | Technical files labels IFUs committee-ready | Local submission tracking attendance | 5–15 business days per pack |
| Launch training | Surgeon and nurse workshops EN FR videos | Venue attendees refresher plan | Within 30 days of appointment |
| Instrument readiness | Tray checklists sterilization guidance maintenance | CSSD capacity maintenance schedule | Pre-launch + quarterly |
| Inventory policy | Stock matrix reorder points consignment options | Safety stock monthly reporting | At contract monthly review |
| Field support | Case shadowing remote and in person troubleshooting | Case scheduling structured feedback | First 3–6 months |
| Country copy | Bilingual brochures listing text FAQs | Localization response SLAs | Live at launch quarterly updates |
Logistics should be predictable in the best sense. Stage fast movers in country—mid sizes and common insert thicknesses—and rotate long-tail sizes from regional hubs. Tie reorder points to case cadence, not wishful thinking. Publish those truths in proposals and committee briefs. Over time, this steadiness becomes your brand.
Documentation is a living asset. Expect periodic audits and label adjustments by market. We keep version-controlled masters so updates flow from factory to distributor to hospital without confusion. Your regulatory manager will have a named counterpart at Ortonom—old-fashioned accountability that saves modern time.
Service closes the loop. Fast instrument turnaround, clear incident paths, and check-ins after early cases turn first wins into durable loyalty. In competitive reviews, reliability often beats a small price edge—especially when the discussion is about surgical instruments for arthroplasty that teams use every day.
Ninety-Day Launch Plan
Prepare weeks 1–3. Finalize bilingual training decks, IFUs, and the spec snapshot table. Confirm regulatory bundles per country and pre-clear customs documents where possible. Build a starter stock matrix aligned to expected case cadence. Choose two lighthouse hospitals and lock dates for workshops and first supervised cases. Position your message clearly around surgical instruments for arthroplasty that are teachable, documented, and deliverable.
Train weeks 4–6. Run workshops for surgeons and scrub teams; schedule and support first cases. Emphasize verification steps that protect results: distal resection checks, tibial slope and rotation, trial tracking with patella. Capture preferences and update the matrix and checklists accordingly.
Execute weeks 7–10. Complete ten to twelve cases per site. Debrief with teams, adjust inventory, and tune instrument maintenance with CSSD. Publish honest lead times and stick to them. If a logistics lane slows, inform committees early and propose a workaround.
Scale weeks 11–13. Add a second country in the same language corridor to reuse training and documents. Approach public tenders once private references are documented. Present outcomes summaries, stable SLAs, and a clear after-sales plan. This blend of tradition and transparency wins tenders and sustains reorders—exactly what matters for surgical instruments for arthroplasty programs.
Partner Invitation
If you bring hospital access and a service-first culture, Ortonom Medical will bring disciplined manufacturing, bilingual enablement, and the surgical instruments for arthroplasty your teams can trust. Let’s schedule the first workshops, run the first cases, and build references that open doors across Southern Africa. That is how we grow—quietly, consistently, and with pride in workmanship—one successful procedure at a time.




 Ortonomy refers to the study and application of principles related to the proper function and structure of systems, especially in biological or technological contexts. In medical terms, it often refers to the alignment and balance of body structures.
Ortonomy refers to the study and application of principles related to the proper function and structure of systems, especially in biological or technological contexts. In medical terms, it often refers to the alignment and balance of body structures.